Designing Hybrids Targeting the Cholinergic System by Modulating the Muscarinic and Nicotinic Receptors: A Concept to Treat Alzheimer's Disease. - Abstract - Europe PMC
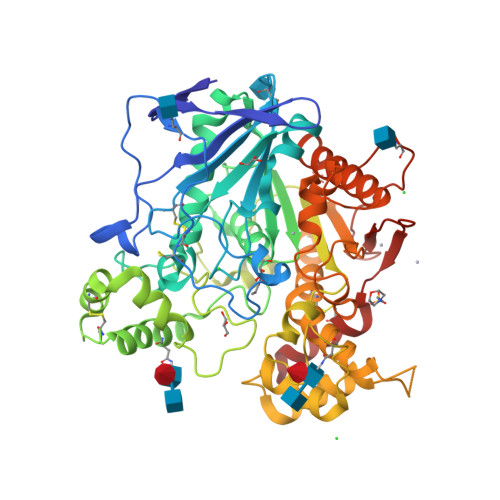
RCSB PDB - 7BO4: Human Butyrylcholinesterase in complex with 3-(2-(butyl(2 -cycloheptylethyl)amino)ethyl)-1H-indol-6-ol

Molecules | Free Full-Text | Designing Hybrids Targeting the Cholinergic System by Modulating the Muscarinic and Nicotinic Receptors: A Concept to Treat Alzheimer's Disease | HTML

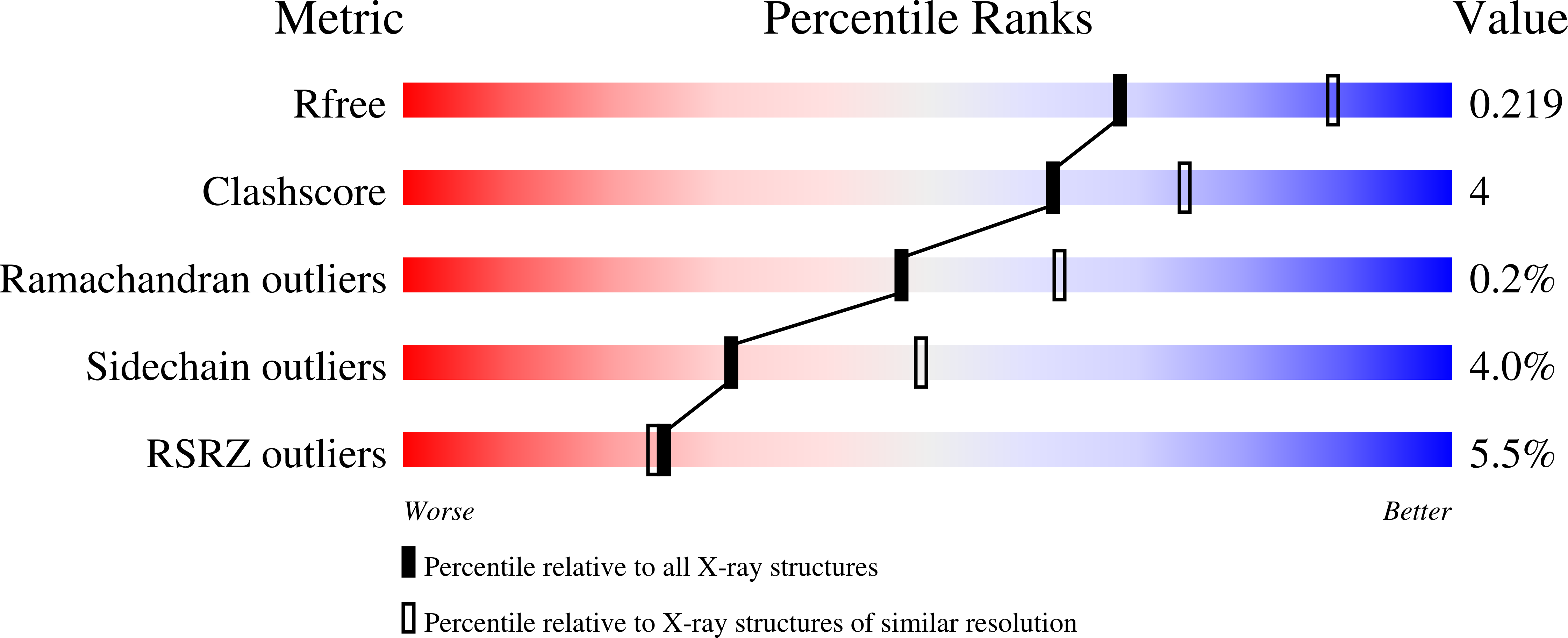
RCSB PDB - 4TPK: Human butyrylcholinesterase in complex with N-((1-(2,3 -dihydro-1H-inden-2-yl)piperidin-3-yl)methyl)-N-(2-methoxyethyl)-2 -naphthamide
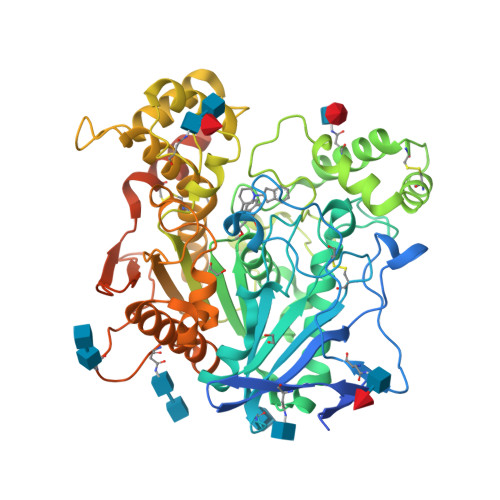
RCSB PDB - 4TPK: Human butyrylcholinesterase in complex with N-((1-(2,3 -dihydro-1H-inden-2-yl)piperidin-3-yl)methyl)-N-(2-methoxyethyl)-2 -naphthamide
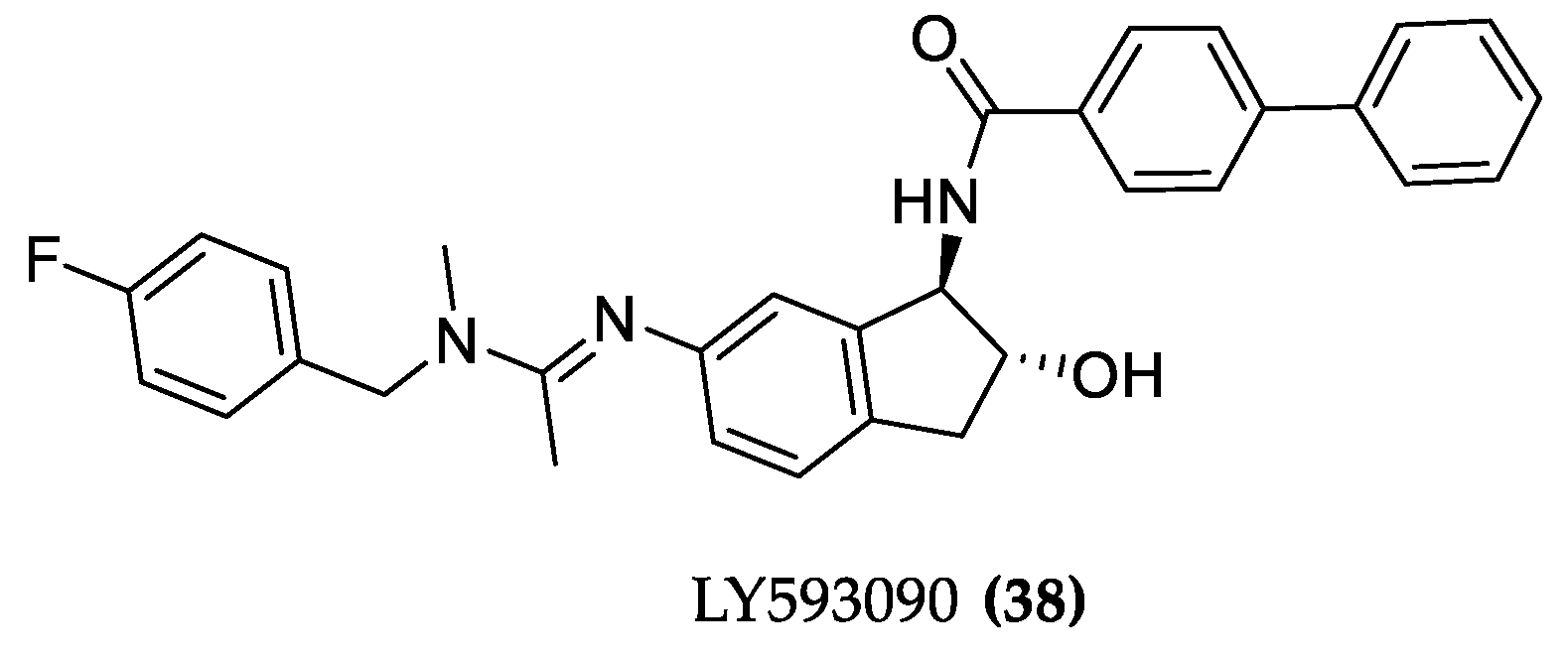
Molecules | Free Full-Text | Designing Hybrids Targeting the Cholinergic System by Modulating the Muscarinic and Nicotinic Receptors: A Concept to Treat Alzheimer's Disease | HTML
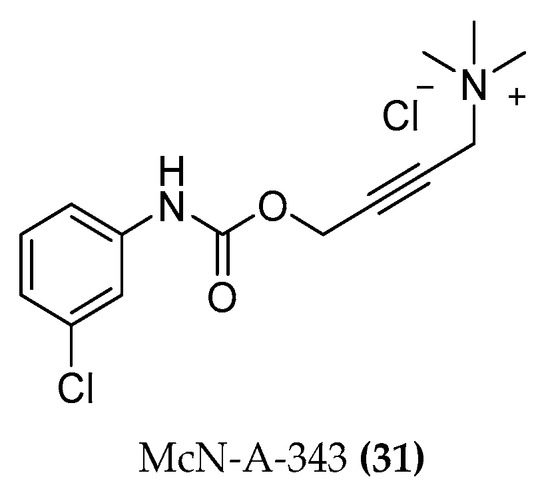
Molecules | Free Full-Text | Designing Hybrids Targeting the Cholinergic System by Modulating the Muscarinic and Nicotinic Receptors: A Concept to Treat Alzheimer's Disease | HTML

Synthesis of the ortho/meta/para Isomers of Relevant Pharmaceutical Compounds by Coupling a Sonogashira Reaction with a Regioselective Hydration | ACS Catalysis

Catalogue International Film Festival Rotterdam 2014 by International Film Festival Rotterdam - Issuu
RCSB PDB - 7BO4: Human Butyrylcholinesterase in complex with 3-(2-(butyl(2 -cycloheptylethyl)amino)ethyl)-1H-indol-6-ol
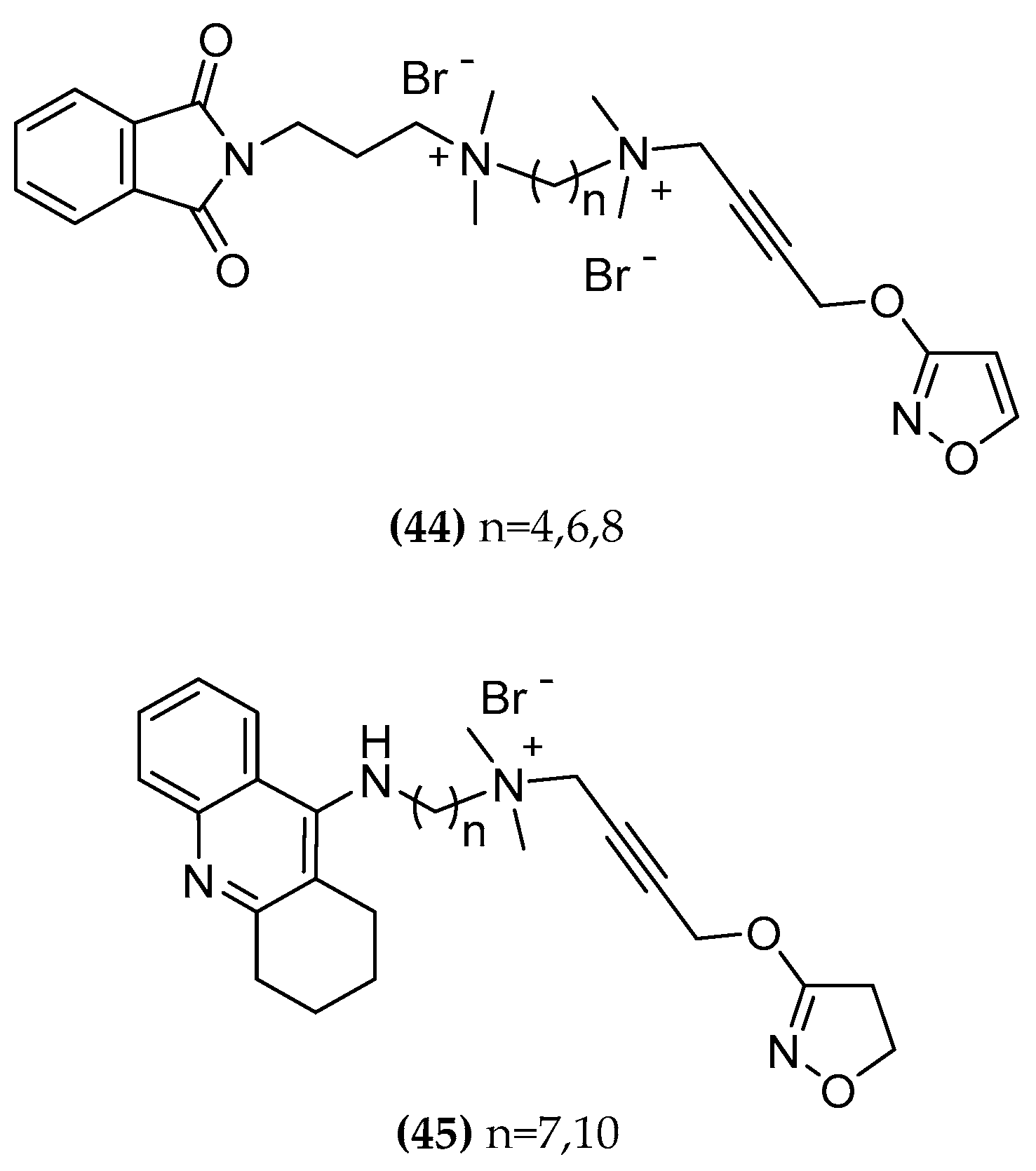
Molecules | Free Full-Text | Designing Hybrids Targeting the Cholinergic System by Modulating the Muscarinic and Nicotinic Receptors: A Concept to Treat Alzheimer's Disease | HTML